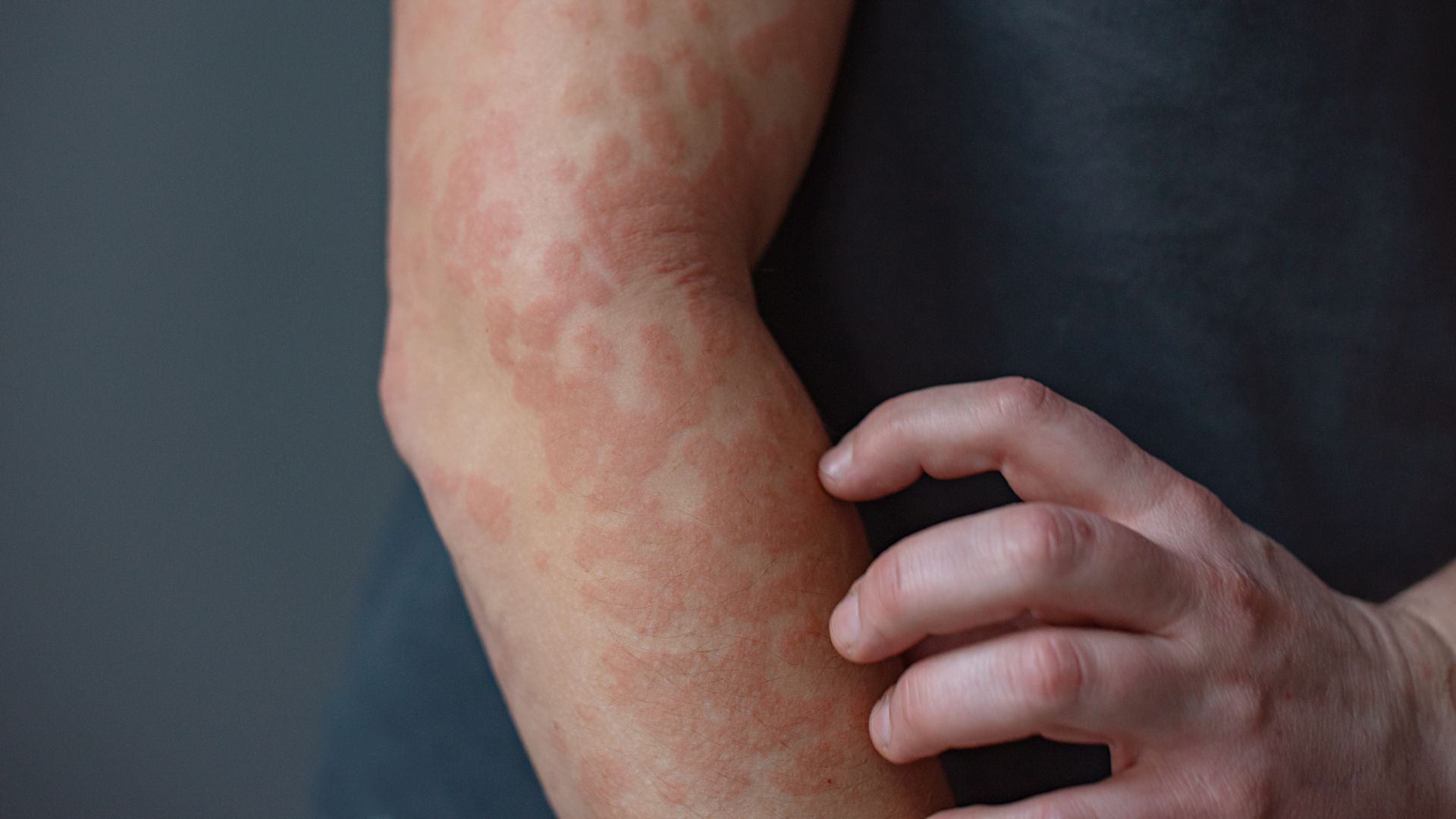
Keratin proteins form our skin, hair and nails — but when mutated, they can mess with the immune system, driving diseases like psoriasis and eczema, new research suggests.
In the new study, published Wednesday (April 8) in the journal Science Translational Medicine, researchers identified a mutant keratin protein that can disrupt processes that normally help skin cells manage inflammation. That breakdown could help to explain how inflammatory skin diseases emerge.
Keratin’s role in skin inflammation
The researchers focused on PC because the genetic disorder is known to be caused by mutations in the KRT16 gene, which carries instructions for keratin 16.
In PC, those mutations disrupt the network of filaments that helps skin cells in high-friction areas of the body, like the soles of the feet, resist mechanical stress. As a result, activities like walking or running cause skin cells to break down and become inflamed, forming painful calluses and blisters.
In people without PC, skin boosts its production of keratin 16 when facing stressors, such as inflammation.
“Keratins are like the steel cables or steel beams of the cell,” Wendy Bollag, a professor in the Augusta University’s department of physiology who wasn’t involved with the study, told Live Science. “That tensile strength you need for the skin, [without it] it’s prone to breaks, tears. Microorganisms can slip in and that will then cause the immune system to react and trigger inflammation.”
And in people with chronic inflammation of the skin, the tissue continually makes lots of keratin 16.
“Understanding why these specific keratin proteins spike during inflammation and directly cause disease when mutated has the potential to identify new ways to treat PC, psoriasis and related conditions,” Cohen and Coulombe said.
So Cohen, Coulombe and colleagues aimed to uncover the mechanism between keratin 16 and PC. They looked at samples of thickened skin from patients with PC, as well as samples from lab mice with a similar condition. They also disabled the KRT16 gene in a second group of mice to see what would happen if the gene didn’t work at all.
They found that when the keratin 16 gene was mutated or missing, skin inflammation skyrocketed. According to Cohen and Coulombe, these results suggested that “K16 normally acts as a ‘brake’ on the signals produced by skin cells to recruit the immune system.”
These signals included type I interferons, a family of proteins that help control inflammation and orchestrate immune responses against viruses and cancer.
In both patients and mice with PC, type I interferon signaling was more active than in people and mice without the condition, the researchers found. The same increase was seen in the mice with no keratin 16, suggesting that when the protein is absent, interferons activate a robust immune response and more inflammation.
On the flip side, when keratin 16 was present, these interferons’ activity was modulated and the inflammation lessened.
Historically, keratin 16 has been “best known as a structural protein,” Cohen and Coulombe said. Learning about this other function “completely changes how we view the skin’s defense system and the role keratins play within it,” they said.
Bollag noted that the study seemed thorough and its results compelling.
“This article is even more impressive than I thought in terms of all of the different techniques and approaches that were used to show that keratin 16 regulates interferon signaling in the skin,” she said. “If you use multiple different techniques and multiple different approaches and they all confirm the same thing, that’s a really powerful way to show that what you’re looking at is real.”
The study authors are optimistic that their findings could have big implications for future treatments for skin diseases. In fact, in the study, they demonstrated that an inhibitor of type I interferons helped clear up skin lesions in a mouse model of PC.
“Understanding this newly found connection creates an opportunity to examine type I interferon signaling as a new therapeutic target in PC,” they said.
This article is for informational purposes only and not meant to offer medical advice.
Cohen, E., Xu, Y., Ghodke, S., Orosco, A., Wang, D., Johnson, C. N., Steen, K., Sarkar, M. K., Özlü, N., Tsoi, L. C., Gudjonsson, J. E., Marchal, L., Hovnanian, A., Parent, C. A., & Coulombe, P. A. (2026). Keratin 16 inhibits type I interferon responses in differentiating keratinocytes of stressed and diseased skin. Science Translational Medicine, 18, eadx9123. 10.1126/scitranslmed.adx9123